Product Details
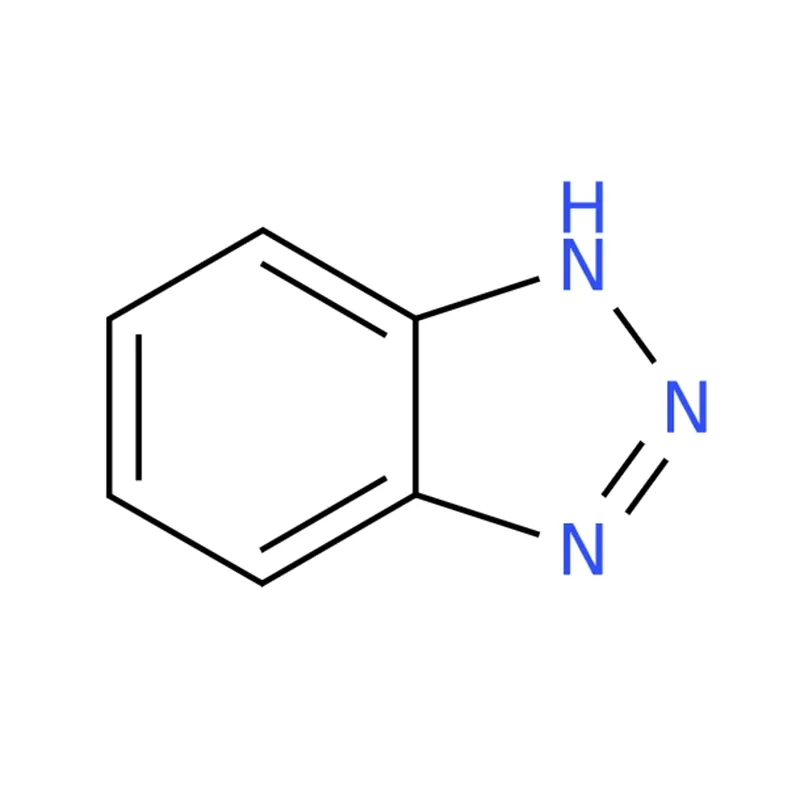
1H-Benzotriazole is the simplest member of the benzotriazole family, featuring a benzene ring fused with a 1H-1,2,3-triazole structure. It acts as both an environmental contaminant and a xenobiotic substance.

Parameters
| Melting point | 97-99 °C(lit.) |
| Boiling point | 204 °C (15 mmHg) |
| density | 1,36 g/cm3 |
| bulk density | 500kg/m3 |
| vapor density | 4.1 (vs air) |
| vapor pressure | 0.04 mm Hg ( 20 °C) |
| refractive index | 1.5589 (estimate) |
| Fp | 170 °C |
| storage temp. | Store below +30°C. |
| solubility | 19g/l |
| form | Powder, Granules, Crystals, Needles or Flakes |
| pka | 1.6(at 20℃) |
| color | White to yellow-beige |
| Odor | Slight characteristic odor |
| PH | 6.0-7.0 (100g/l, H2O, 20℃)suspension |
| explosive limit | 2% |
| Water Solubility | 25 g/l in water (20 ºC) |
| Merck | 14,1108 |
| BRN | 112133 |
| Stability: | Stable, but may be light sensitive. Incompatible with strong oxidizing agents, heavy metals. |
| InChIKey | QRUDEWIWKLJBPS-UHFFFAOYSA-N |
| LogP | 1.34 at 22.7℃ |
| CAS DataBase Reference | 95-14-7(CAS DataBase Reference) |
| NIST Chemistry Reference | 1H-Benzotriazole(95-14-7) |
| EPA Substance Registry System | 1,2,3-Benzotriazole (95-14-7) |
Safety Information
| Hazard Codes | Xn,Xi,F |
| Risk Statements | 20/22-36-52/53-5-36/37/38-20/21/22-11 |
| Safety Statements | 26-36/37-61-45-36/37/39-28A |
| RIDADR | 2811 |
| WGK Germany | 1 |
| RTECS | DM1225000 |
| Autoignition Temperature | 400 °C |
| Hazard Note | Harmful/Irritant |
| TSCA | Yes |
| HS Code | 29339990 |
| Hazardous Substances Data | 95-14-7(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 560 mg/kg LD50 dermal Rabbit > 2000 mg/kg |
1H-Benzotriazole is a highly effective corrosion inhibitor, widely used in aircraft deicing and antifreeze fluids. It is also commonly incorporated into dishwasher detergents. Furthermore, it acts as a restraining agent in photographic emulsions and serves as a standard analytical reagent for the determination of silver. As a corrosion inhibitor, it demonstrates excellent protective performance both in atmospheric environments and under water. In addition, it plays an important role in the synthesis of amines from glyoxal.