Product Details
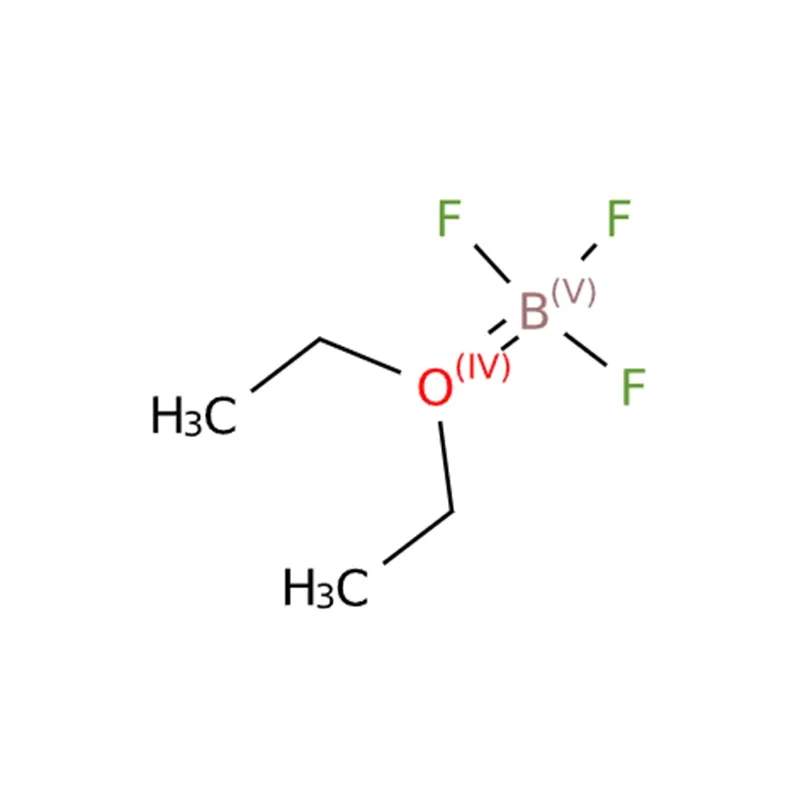
Product Description of Boron Trifluoride Diethyl Etherate CAS#109-63-7
Boron trifluoride is a key Lewis acid catalyst in organic synthesis, widely involved in reactions such as polymerization, hydrocarbon processing, alkylation, and isomerization. Due to its difficulty in handling in gaseous form, it is commonly combined with polar oxygen- or nitrogen-containing compounds to form stable complexes, which can meet the activity requirements of various reactions. Boron trifluoride diethyl etherate is one of the most widely used complexes among these. Additionally, boron trifluoride can readily form complexes with thioethers, ketones, anisole, phenol, amines, and many of their derivatives, further expanding its applicability in organic synthesis.

Boron trifluoride diethyl etherate Chemical Properties
| Melting point | −58 °C(lit.) |
| Boiling point | 126-129 °C(lit.) |
| Density | 1.15 g/mL(lit.) |
| Vapor density | 4.9 (vs air) |
| Vapor pressure | 4.2 mm Hg ( 20 °C) |
| Refractive index | n20/D 1.344(lit.) |
| Fp | 118 °F |
| Storage temp. | Store below +30°C. |
| Solubility | Miscible with ether and alcohol. |
| Form | liquid |
| Specific Gravity | 1.126 (20/4℃) |
| Color | brown |
| Explosive limit | 5.1-18.2%(V) |
| Water Solubility | Reacts |
| Sensitive | Moisture Sensitive |
| Hydrolytic Sensitivity | 7: reacts slowly with moisture/water |
| Merck | 141,350 |
| BRN | 3909607 |
| Exposure limits | ACGIH: TWA 0.1 ppm; Ceiling 0.7 ppm |
| Stability | Stable. Highly flammable. May form explosive peroxides in contact with air or oxygen. Reacts exothermically with water to form extremely flammable diethyl ether and toxic, corrosive boron trifluoride hydrates. Also incompatible with bases, amines, alkali metals. |
| InChIKey | MZTVMRUDEFSYGQ-UHFFFAOYSA-N |
| CAS DataBase Reference | 109-63-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Boron trifluoride etherate(109-63-7) |
| EPA Substance Registry System | Boron, trifluoro[1,1'-oxybis[ethane]]-, (T-4)- (109-63-7) |
| Hazard Codes | T,C |
| Risk Statements | 10-14-20/22-35-48/23-34-14/15-23-22 |
| Safety Statements | 16-23-26-36/37/39-45-8-28A-43 |
| RIDADR | UN 2604 8/PG 1 |
| WGK Germany | 3 |
| F | 10 |
| Autoignition Temperature | 185 °C DIN 51794 |
| TSCA | Yes |
| HazardClass | 8 |
| PackingGroup | I |
| HS Code | 29319090 |
| Hazardous Substances Data | 109-63-7(Hazardous Substances Data) |
Product Application of Boron Trifluoride Etherate (CAS#109-63-7)
Boron trifluoride etherate (CAS#109-63-7) is widely applied in multiple industrial and chemical synthesis fields, with its core value reflected in serving as a high-efficiency catalyst and key raw material. It is extensively used as a cationic polymerization catalyst in the production of butadiene rubber and polyformaldehyde, effectively promoting the polymerization reaction, improving product yield and quality. As a key raw material, it plays an important role in the preparation of boron–hydrogen high-energy fuels, providing essential support for the development of high-energy materials. Meanwhile, it is also widely used in the extraction of boron isotopes, especially in the isolation and purification of boron-10 isotope, which is crucial for related scientific research and industrial production. In chemical synthesis, it is commonly used as a catalyst to promote various organic reactions, and also serves as an important starting material for the synthesis of boron–hydrogen high-energy dyes, further expanding its application scope in fine chemical industry. Its stable catalytic performance and wide adaptability make it an indispensable material in organic synthesis, polymer production, and isotope separation fields.