Product Details
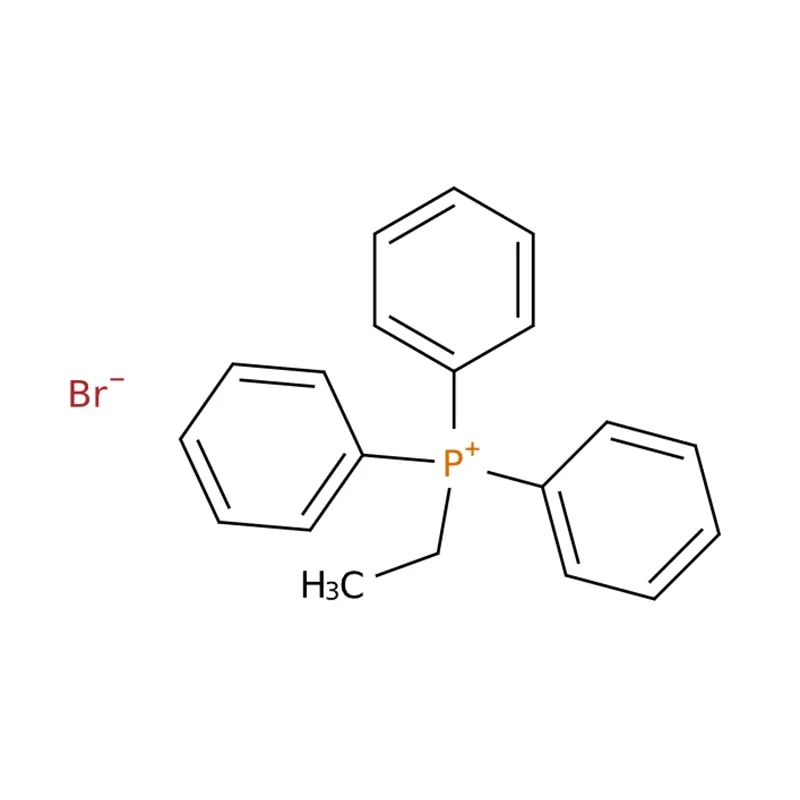
Ethyltriphenylphosphonium bromide generally presents as a white or off-white crystalline powder. It is soluble in organic solvents including ethanol, chloroform and dichloromethane, and shows limited solubility in water. This compound can be used as a dyeing reagent, and similarly to other phosphorus salts, it also exhibits antiviral activity.
| Product Name: | Ethyltriphenylphosphonium bromide |
| Synonyms: | Ethyltriphenylbromophosphine;ethyltriphenyl-phosphoniubromide;Ethyl(trisphenyl)phosphonium bromide;Ethyl triphenyl phosphoniuM broMide (ETPB);TEP (oniuM coMpound);ETHYLTRIPHENYLPHOSPHONIUM BRIMIDE;ethyl triphenylphosphoiuum bromide;ETHYLTRIPHENYLPHOSPHONIUM BROMIDE 99+% |
| CAS: | 1530-32-1 |
| MF: | C20H20BrP |
| MW: | 371.25 |
| EINECS: | 216-223-3 |
| Product Categories: | Phosphonium SaltsC-C Bond Formation;Greener Alternatives: Catalysis;C-C Bond Formation;Olefination;Wittig Reagents;Phase Transfer Catalysts;Phosphonium Compounds;Synthetic Organic Chemistry;Wittig & Horner-Emmons Reaction;Wittig Reaction;organophosphorus compound |
| Mol File: | 1530-32-1.mol |

Ethyltriphenylphosphonium bromide Chemical Properties
| Melting point | 203-205 °C(lit.) |
| Boiling point | 240℃[at 101 325 Pa] |
| Density | 1.38[at 20℃] |
| Vapor pressure | 0-0.1Pa at 20-25℃ |
| Fp | 200 °C |
| Storage temp | Inert atmosphere,Room Temperature |
| Solubility | 174g/l soluble |
| Form | Crystalline Powder |
| Color | White to off-white |
| Water Solubility | 120 g/L (23 ºC) |
| Sensitive | Hygroscopic |
| BRN | 3599630 |
| InChIKey | JHYNXXDQQHTCHJ-UHFFFAOYSA-M |
| LogP | -0.69--0.446 at 35℃ |
| CAS DataBase Reference | 1530-32-1(CAS DataBase Reference) |
| EPA Substance Registry System | Phosphonium, ethyltriphenyl-, bromide (1530-32-1) |
Ethyltriphenylphosphonium Bromide (ETPB) serves as a phase-transfer catalyst, which accelerates the curing of phenolic epoxy resins, partial fluoroelastomer resins and thermosetting powder coatings. It also acts as a catalyst for the synthesis of various organic compounds and functions as a pharmaceutical intermediate.
ETPB can be used as a reactant in the synthetic routes: preparing D-amino acids from L-cysteine-derived thiazolidines, synthesizing Leiodolide A via aldol reactions, and participating in Horner–Wadsworth–Emmons olefination.
Besides, it is applicable to the preparation of cycloalkanoindolines through diastereoselective intramolecular imino-ene reactions.
It is also adopted as a reagent in solid-state metathesis polycondensation for the preparation of alkyl-dipropenylthiophene monomers, as well as in Mizoroki–Heck cyclization and cascade Tsuji–Trost cyclization reactions.