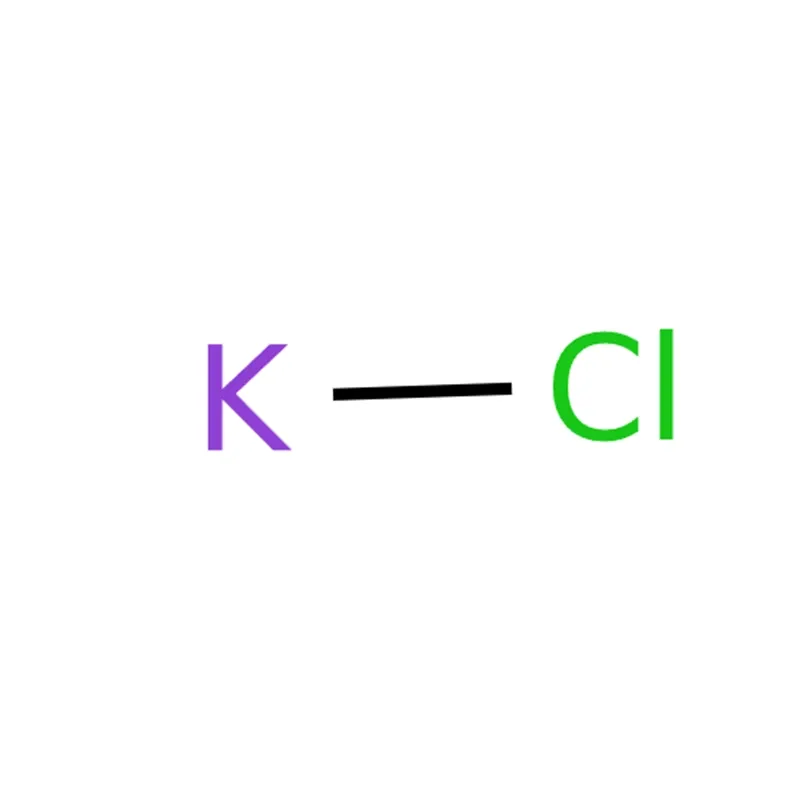
Potassium Chloride CAS# 7447-40-7
Potassium chloride (KCl) is a metal halide salt featuring extensive application coverage. It is primarily applied as fertilizer, supplying essential potassium nutrients for plant growth and strengthening crop disease resistance. It also sees wide usage across the food and pharmaceutical industries. In medical fields, potassium chloride tablets are commonly adopted to treat hypokalemia by restoring and maintaining normal blood potassium levels. Within food processing, it acts as an electrolyte replenisher, functional salt substitute, and firming agent to optimize food texture and structural stability.

Potassium Chloride Chemical Properties
| Melting point | 770 °C (lit.) |
| Boiling point | 1420°C |
| Density | 1.98 g/mL at 25 °C (lit.) |
| Bulk density | 1000kg/m3 |
| Refractive index | n20/D 1.334 |
| Fp | 1500°C |
| Storage temp. | 2-8°C |
| Solubility | H2O: soluble |
| Form | random crystals |
| Color | White |
| Specific Gravity | 1.984 |
| Odor | Odorless |
| PH Range | 7 |
| Flame Color | Light Purple |
| PH | 5.5-8.0 (20℃, 50mg/mL in H2O) |
| Biological source | rabbit |
| Water Solubility | 340 g/L (20 ºC) |
| Sensitive | Hygroscopic |
| λmax | λ: 260 nm Amax: 0.02 |
| λ: 280 nm Amax: 0.01 | |
| Crystal Structure | NaCl type |
| crystal system | Cube |
| Merck | 147,621 |
| Sublimation | 1500 ºC |
| BRN | 1711999 |
| Space group | Fm3m |
| Lattice constant | a/nmb/nmc/nmα/oβ/oγ/oV/nm30.62930.62930.62939090900.2492 |
| Dielectric constant | 4.6(Ambient) |
| BCS Class | 1 |
| Stability | Stable. Incompatible with strong oxidizing agents, strong acids. Protect from moisture. Hygroscopic. |
| InChIKey | WCUXLLCKKVVCTQ-UHFFFAOYSA-M |
| CAS DataBase Reference | 7447-40-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Potassium chloride(7447-40-7) |
| EPA Substance Registry System | Potassium chloride (7447-40-7) |
Product Usage
Potassium chloride (KCl) is widely used in pharmaceutical formulations, food additives, and as a chemical reagent. Replacing table salt (sodium chloride) with potassium chloride can help lower dietary sodium intake and may offer health benefits. In addition, molten potassium chloride is utilized in the electrolytic production of metallic potassium. KCl is naturally present in seawater brine and can also be obtained through extraction from the mineral carnallite.