Product Details
Product Description of Sodium Oleate CAS#143-19-1
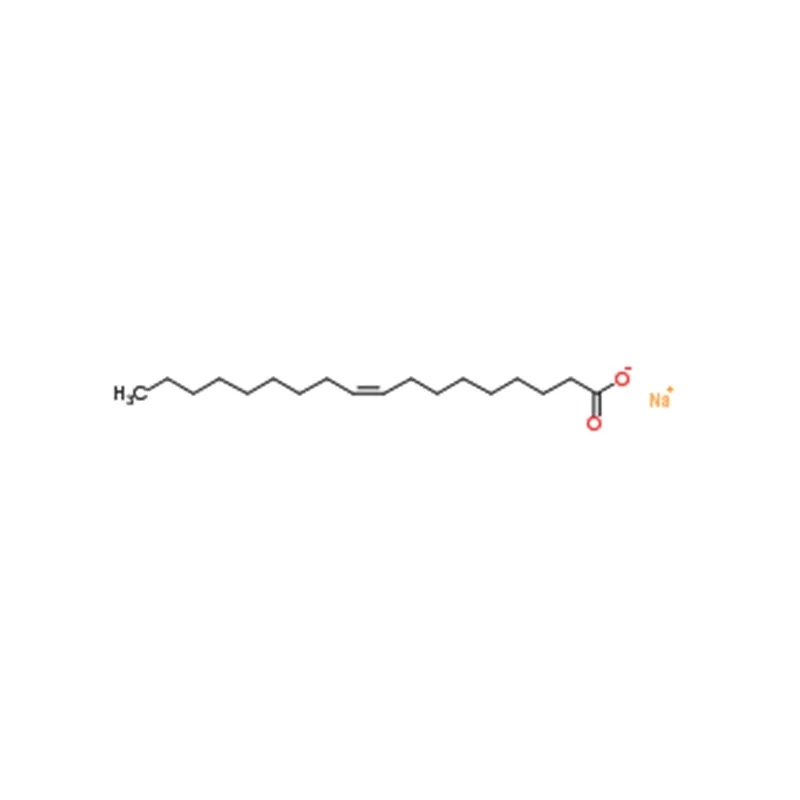
Sodium oleate, also referred to as sodium octadecenoate, is an organic compound with the chemical formula C₁₇H₃₃CO₂Na. It constitutes the main ingredient of soaps made from olive oil and tallow, and can also be synthesized via the reaction between sodium hydroxide and oleic acid.
As an amphiphilic substance with both hydrophobic and hydrophilic segments, sodium oleate shows superior emulsifying, penetrating and cleaning capabilities. It dissolves easily in hot water and is extensively employed as an anionic surfactant, along with uses including fabric waterproofing treatment.

Sodium oleate Chemical Properties
| Melting point | 232-235 °C (lit.) |
| storage temp. | -20°C |
| solubility | Methanol (Slightly) |
| form | Powder |
| color | White to slightly yellow |
| Odor | slt tallow odor |
| Water Solubility | soluble H2O, partially decomposes; soluble alcohol [HAW93] |
| Merck | 146828 |
| BRN | 4046357 |
| Hydrophilic-Lipophilic Balance (HLB) | 18 |
| Dielectric constant | 2.7(20℃) |
| Stability: | Stable. Incompatible with strong oxidizing agents. |
| InChI | InChI=1S/C18H34O2.Na/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18(19)20;/h9-10H,2-8,11-17H2,1H3,(H,19,20);/q;+1/p-1/b10-9-; |
| InChIKey | BCKXLBQYZLBQEK-KVVVOXFISA-M |
| SMILES | C(CCCCCC([O-])=O)C/C=C\CCCCCCCC.[Na+] |
| LogP | 7.698 (est) |
| CAS DataBase Reference | 143-19-1(CAS DataBase Reference) |
| EPA Substance Registry System | Sodium oleate (143-19-1) |
| Safety Statements | 22-24/25-25-24 |
| WGK Germany | 1 |
| RTECS | RK1200000 |
| F | 46318 |
| HS Code | 29161500 |
| Hazardous Substances Data | 143-19-1(Hazardous Substances Data) |
| Toxicity | LD50 ivn-mus: 152 mg/kg RPOBAR 2,327,70 |
Product Application of Sodium Oleate CAS#143-19-1
Chemically pure sodium oleate is extensively used in mineral processing, fabric waterproofing, and soap production. It also acts as an anionic surfactant and a waterproofing agent for textile materials. Analytical reagent (AR) grade sodium oleate is applied in biochemical studies, for instance, in the activation of protein kinase C in hepatic cells.